 "σ-Bond Metathesis: A 30-Year Retrospective". Here two pi bonds can exist between two atoms. During the bonding between two given atoms, Only one sigma bond is formed. Pi bonds between two atoms are formed after sigma bonds are formed between them. Sigma bonds are formed first when atoms interact. It has been suggested that dehydrocoupling reactions proceed via sigma-bond metathesis. Pi bonds are usually weaker compared to sigma bonds. Unfortunately the reaction does not readily allow the introduction of functional groups. 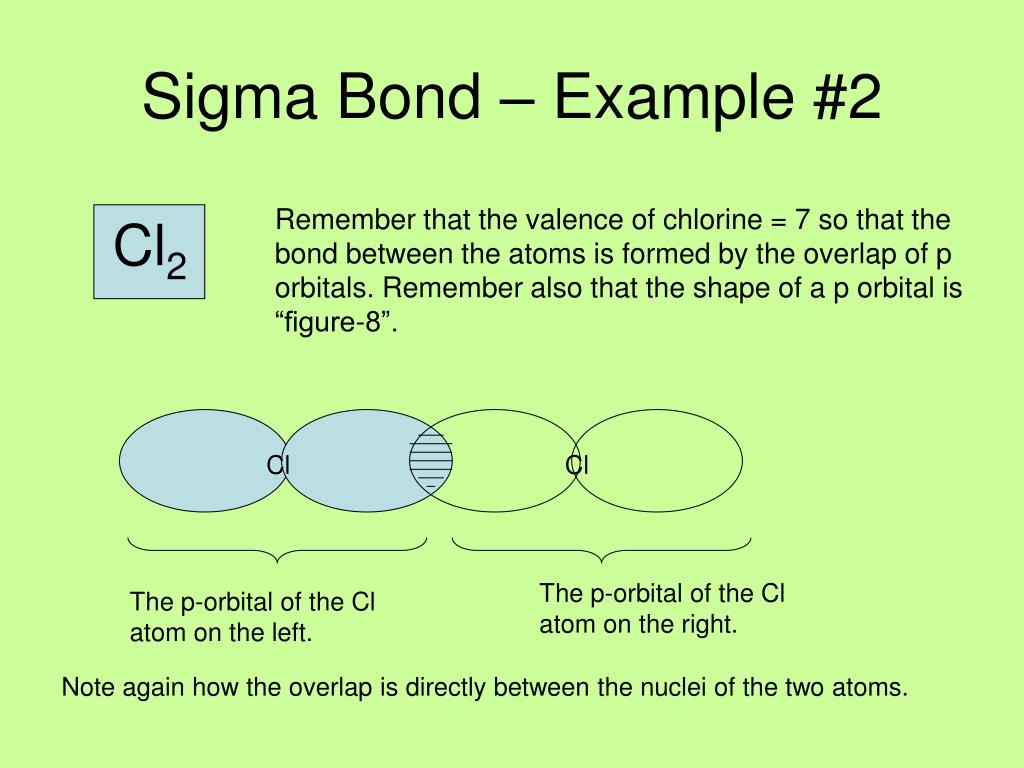
The reaction attracted much attention because hydrocarbons are normally unreactive substrates, whereas some sigma-bond metatheses are facile. In sigma bond ,the electron cloud is symmetrical about the internuclear axis and in π bond, it is unsymmetrical, being more concentrated above and below the plane of atomic nuclei.\( \newcommand\): The currently accepted mode for the mechanism of \(\sigma\) bond metathesis involving a kite-like transition state. (3) In case of formation of Pi bond, sideways overlapping take place which is only to do a small extend. 
(2) In the formation of sigma bond overlapping is along the internuclear axis ,therefore ,there is considerable overlapping. 
Ah, Pie Bond, which uses the simple pine, is the weaker form of a Kobelev bond that insulate and protects the Sigma bond. Its the strongest form of a Kobelev bond that directly connects elements together. (1) The strength of a bond depends upon the extent of overlapping. Now, with these types of chemical bonds, we can talk about sigma versus pie bonds now a Sigma bond, which uses the symbol Sigma. In nitrogen molecule the electronic configuration is 1s 2 2s 2 2p x 1 2p y 1 2p z 1 atoms are held together by 1 Sigma and 2 Pi bonds. In a sigma bond, the electron pair occupies an orbital a region of space associated with a particular value of the energy of the systemlocated mainly between the two atoms and symmetrically distributed about the line determined by their nuclei. In oxygen molecule the electronic configuration is 1s 2 2s 2 2p x 2 2p y 1 2p z 1 , the two atoms are held together by one sigma bond and one pi bond. Pi bond is formed by lateral or sideways overlapping of p orbital that is why overlapping of p orbital in a direction at right angles to the inter nuclear axis. If Z axis is chosen as the internuclear axis, no overlapping can take place between s and p x, p y or p z orbital i.e. The molecule of fluorine is produced by overlapping of 2p z orbitals of the two fluorine atoms. This type of overlapping occurs when p-orbital of one atom overlaps with the p orbital of the other. In the formation of hydrogen fluoride, 1s orbital of hydrogen overlaps with 2p z orbital fluorine. Accordingly, sigma bonds are formed between the C and O atoms in the ketone with the corresponding metal atoms of the surface. Half filled s-orbital of one atom overlaps with the half filled p- orbital of another atom. In short, you can remember that single bonds are sigma bonds. 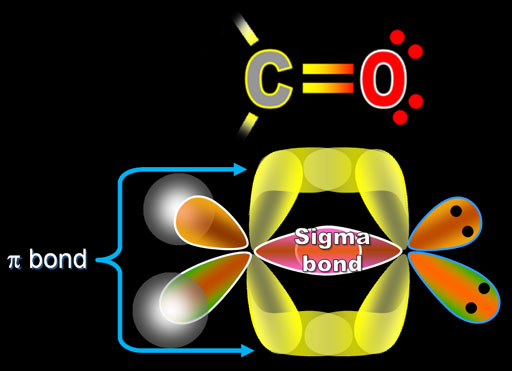
In the formation of hydrogen molecule, 1s orbital of one hydrogen atom overlap with 1s orbital of other hydrogen atom and thus forming a sigma bond. Overlapping that take place between atoms having half-filled s – orbital in their outermost energy shell. The bonds between the sp 3 orbitals of hybridized carbon and the s orbitals of hydrogen in methane are also example of sigma bonds. Then the next pie bond would save a little bit less energy, and then the last pie bond would not save that much energy at all, but it would still be a little bit. The single bond is the strongest because the single bond saves the most energy of the three bonds for the triple. Single bonds in Lewis structures are described as latexsigma/latex bonds in valence bond theory. The overlapping along the internuclear axis can take place in any one of the ways: The answer is that the Sigma Im sorry, the single bond, not Orbital. When a bond is formed between two atoms by the overlap of their atomic orbitals along the internuclear axis, the bond formed is called sigma bond A sigma bond can be formed by overlap of an s atomic orbital with a p atomic orbital. Depending upon the type of overlapping, the covalent bonds are mainly of two types: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
